Aretaeus (130-200) was the first
physician to give diabetes its
proper name. In his treatise 'On
the causes and symptoms of
chronic diseases Book II'. He
used the Greek word diabetes
(meaning siphon) to describe the disease.
He stated that ' Diabetes
is a remarkable affection not very
frequent among men being a
melting down of the flesh and
blood into urine.' Mellitus was
added later by others to denote
the sweet taste of urine. Mellitus
means honey in Greek.
Aretaeus (130-200) was the first
physician to give diabetes its
proper name. In his treatise 'On
the causes and symptoms of
chronic diseases Book II'. He
used the Greek word diabetes
(meaning siphon) to describe the disease.
He stated that ' Diabetes
is a remarkable affection not very
frequent among men being a
melting down of the flesh and
blood into urine.' Mellitus was
added later by others to denote
the sweet taste of urine. Mellitus
means honey in Greek.
 Claude Bernard (1813-1878). The
father of 'experimental physiology'
showed that the liver stores
glycogen which can be converted
into glucose.
Claude Bernard (1813-1878). The
father of 'experimental physiology'
showed that the liver stores
glycogen which can be converted
into glucose.
 Oscar Minkowski (1858-1931)
discovered in the laboratory of
the Medical Clinic of Strassburg
in 1889 removal of pancreas
from dog caused it to develop
severe diabetes with excessive
thirst and polyuria.
Oscar Minkowski (1858-1931)
discovered in the laboratory of
the Medical Clinic of Strassburg
in 1889 removal of pancreas
from dog caused it to develop
severe diabetes with excessive
thirst and polyuria.
 Frederick Banting (1891-1941)
revolutionized the treatment
of diabetes after successfully
isolating insulin from his
dog Marjory in 1921.
Frederick Banting (1891-1941)
revolutionized the treatment
of diabetes after successfully
isolating insulin from his
dog Marjory in 1921.
![]()
(for an enlarged view of the retina please click the picture)
2.1 CLASSIFICATION
Diabetic retinopathy is classified as:
- background retinopathy
- proliferative retinopathy
Each has a different prognosis for vision.
Background diabetic retinopathy (BDR) is further classified as:
- minimal
- moderate
- severe
- very severe
Precise grading for each level of BDR is based on the Airlie House
Grading system (see below) 13. Severe and very severe BDR are
commonly referred to as preproliferative diabetic retinopathy (PDR).
.
BDR which affects the macula is described as:
- diabetic maculopathy
Diabetic maculopathy (DM) is further classified as:
- focal oedema (also known as exudative maculopathy
- diffuse oedema
- ischaemia or
- mixed
location as:
- new vessels on the disc (NVD) or within 1 disc diameter
- new vessels elsewhere in the retina (NVE) (more than
of the disc (DD)
.
1 DD from the disc)
and according to severity as early PDR, established PDR, florid PDR
and gliotic PDR. 'Involutionary' PDR is used to describe new vessels
which have regressed in response to treatment or (rarely)
spontaneously.
.
2.2 BACKGROUND DIABETIC RETiNOPATHY
BDR, also described as non-proliferative diabetic retinopathy (NPDR),
is so-called because the lesions lie within the retina (i.e. they form
a 'background' to lesions on the retinal surface such as new vessels
or within the vitreous such as forward new vessels and vitreous
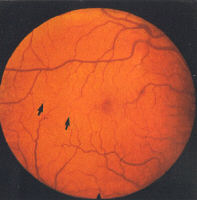
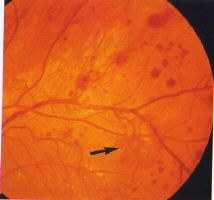
haemorrhage). Initially, background retinopathy consists of micro-
aneurysms only (Fig 1), progressing to microaneurysms and small
haemorrhages ('dots and blots') (Fig 2) which can be graded as mild,
moderate and severe (Fig 3). Splinter haemorrhages
are also seen in combined hypertensive/diabetic retinopathy (Fig 4).
Some grading centres consider a minimum of 4 microaneurysms are
required to diagnose background retinopathy and that they should
be bilateral.
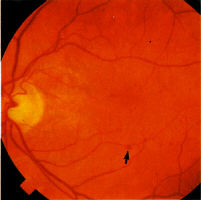
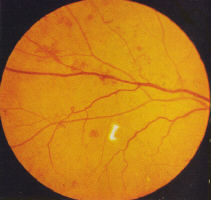
Exudates, more common in NIDDM, are waxy yellow deposits with discrete
edges, extending to the equatorial fundus often in clusters or forming
circinate patterns whose centre may be a leaking microaneurysms (Fig 5).
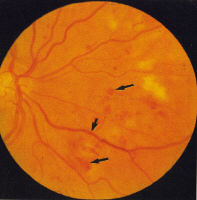
'Cotton wool spots' (CWS), previously called soft exudates, are fluffy
white lesions representing infarcts of the nerve fibre layer hence are only
found in the posterior retina where the nerve fibre layer is of appreciable
thickness (Fig 6). They may appear suddenly during periods of changing
glucose regulation and in association with hypertension.
 |
 |
.
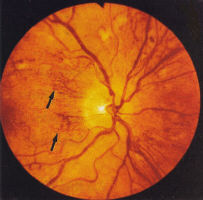
Venous dilatation may occur as an early sign (see Fig 3) but become
more pronounced as more of the capillary bed is closed. A general
dilatation of the veins is observable even when the retinopathy is very
mild and is to be distinguished from venous beading which is a sign of
preproliferative retinopathy (Fig 7). Beading is indicative of extensive
ischaemia of the retina and manifests as saccular bulges in the wall of
the vein (Fig 7). Fluorescein angiography will invariably show closure
of the capillary bed on either side of the vessel (Fig 8).
 |
 |
.
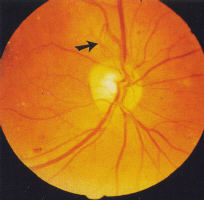
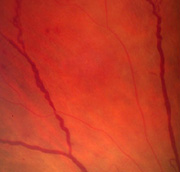
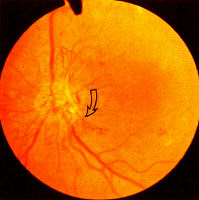
Other signs of preproliferative retinopathy retinopathy are dilated
capillaries which can mimic new vessels but are better described as
intraretinal microvascular abnormalities (IRMA) (see Fig 4). They
frequently occur adjacent to CWS's, and may be associated with
other signs such as 'omega' venous loops (Fig 9), venous reduplication
and white lines which represent occluded arterioles (Fig 10). According
to the Early Treatment for Diabetic Retinopathy Study (ETDRS) report,
preproliferative retinopathy is definitely present if the signs conform to
the 1-2-3 rule: i.e.. the presence of venous beading and/or IRMA and/or
large blot haemorrhages in 1-3 quadrants of the fundus.
.
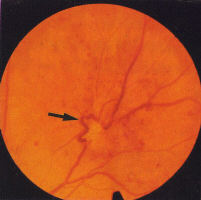 |
 |
.
Precise classifications for the purposes of clinical trials and other
studies based on the Airlie House grading system have used the
following criteria to define each of the grades of BDR:
- mild BDR: at least one microaneurysm
- moderate BDR: severe retinal haemorrhages in at least one
- severe BDR: severe retinal haemorrhages in four quadrants;
- very severe BDR: any two of the features of severe BDR.
quadrant, or CWS, venous beading or IRMA definitely present
'
or venous beading in 2 quadrants; or extensive IRMA in one
quadrant.
.
However, such classifications are difficult to use in clinical practice on a
routine basis since certain features such as venous beading are common
to both moderate and severe retinopathy. It is preferable, therefore to
consider BDR as either midl or 'low risk' (i.e. not requiring regular close
observation by an ophthalmologist) and severe or 'high risk' (i.e. requiring
regular close observation as prelude to panretinal photocoagulation, PRP).
For the purposes of defining those patients at risk of developing new
vessels, the features of 'low risk' (mild) BDR are:
- mildly dilated veins
- microaneurysms
- small haemorrhages
- hard exudates
- occasional CWS's
and the features of 'high risk', preproliferative (severe) BDR are
- IRMA
- venous beading and 'omega' loops
- clusters of large 'blot' or 'blotch' haemorrhages
- multiple CWS's
.
2.3 PROLIFERATIVE DIABETIC RETiNOPATHY (PDR)
Proliferative retinopathy (PDR) usually appears late in the disease.
However, PDR may occur with little warning in young adolescent
or post adolescent individuals and have a particularly aggressive
course. These individuals require extensive counselling (see below).
It is important to emphasize that new vessels by themselves rarely
produce symptoms; their sequelae are the cause of visual loss.
New Vessels mostly arise from the venous side of the circulation
and are recognizable by their abnormal location and their unusual
pattern (Fig 11). Unlike normal vessels which have a branching
pattern that divides dichotomously, new vessels form loops or rete
(arcades). While norm vessels appear to be supplying or draining an
area of retina, it may be difficult to identify such a role for new
vessels. For example, a rete of vessels may arise from the main trunk
of a vein and criss-cross the vessel randomly or a venule may arise
from the disc and after forming a tortuous loop wind back towards
the disc (Fig 12, 13). Early new vessels usually lie flat on the surface
of the retina, but when the vitreous is detached, they are drawn
forward as a result of vitreous traction.
.
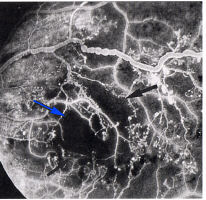
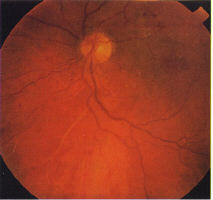
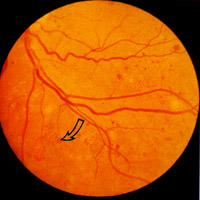
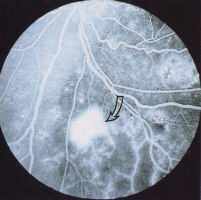
New vessels arise from the disc (NVD) (Fig 13, 14, 15) or the retina
(NVE) (Fig 11, 12). Most new vessels arise from veins in a central,
circular areas about 3½ disc diameter from the disc margin in any of
4 quadrants. Where there is sectoral ischaemia, new vessels
characteristically arise at the junction of the perfused and non-
perfused area as demonstrated on fluorescein angiograms (Fig 16a, b).
In eyes with widespread ischaemia, NVD are common. Disc NVD may
be flat or forward depending on the position of the posterior vitreous
face.
.
 |
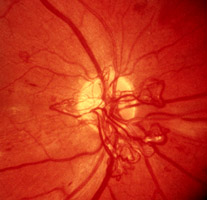 |
 ........... ........... |
.
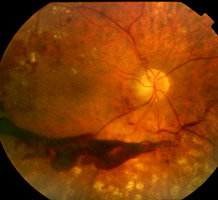
Untreated NV (D or E) lead to vitreous haemorrhage (VH) and blindness.
VH may be subhyaloid before the vitreous is fully detached and present
as small 'crescents' with a level superior border if the PVD is present
(Fig 17, 18).
.
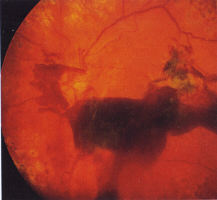
In the late stage of the natural history of proliferative disease, fibrous
tissue (gliosis) gathers around the new vessels and contraction of this
tissue causes repeated bleeding and eventually, tractional retinal
detachment. Occasionally, fine epiretinal gliosis occurs with minimal
traction on the retina but this is uncommon.
.
 |
 |
2.4 MACULOPATHY
Visual loss in diabetic maculopathy is usually the result of macular
oedema. Macular oedema may be difficult to detect; characteristically
it appears as 'retinal thickening' on binocular, stereoscopic slit lamp
examination. When it occurs within one disc diameter of the fovea, it is
termed clinically significant macular oedema (CSME)14 since under these
conditions it is visually threatening. Fluorescein angiography also reveals
vessels with abnormal permeability causing leakage and pooling of dye in
the late phase. A classification of maculopathy based on ophthalmological
features is detailed below. However it is important to note than the terms
'focal' and 'diffuse' are used to convey a sense of the extent of macular
involvement, and if the ophthalmological findings are interpreted in
conjunction with the corrected visual acuity a more accurate impression
of the severity of disease will be obtained. For instance, diabetic
maculopathy may occur in the relative absence CSME and fluorescein
leakage on angiography. In this condition, ischaemia is the likely pathology.
.
2.4.1 Clinical types of diabetic maculopathy
2.4.1.1 Focal maculopathy
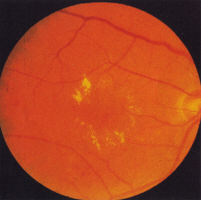
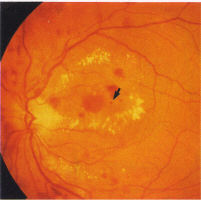
The characteristic features of focal maculopathy are well circumscribed,
leaking areas associated with complete or incomplete rings of hard exudates.
These are often related to microaneurysms, particularly in the centre of
exudative rings (Fig 5, 19). The exudative rings have a predilection for the
perifoveal area where the retina is thickest. The focal areas of leakage are
thickened by retinal oedema and fluorescein angiography is usually not
necessary to identify them. Recent studies also suggest that retinal
pigment epithelial damage may contribute to the macular oedema probably
by failing to remove the tissue fluid accumulating in the retina from the
leaking capillaries15.
.
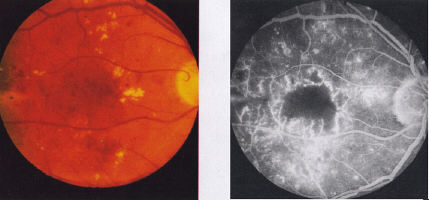
2.4.1.2 Diffuse maculopathy
Diffuse maculopathy consists of generalized leakage from dilated capillaries
in the macular area. Severe oedema is a feature and it is often associated
with cystic changes. The other features of diabetic retinopathy may not be
present and in particular there may be no exudates. In severe cases it may
be impossible to identify the fovea due to the diffuse retinal thickening. The
fluorescein angiogram is more dramatic than the ophthalmoscopic picture
(FIg 20 a, b).
 |
|
|
.
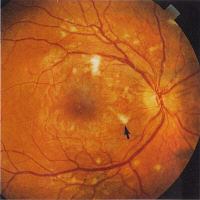
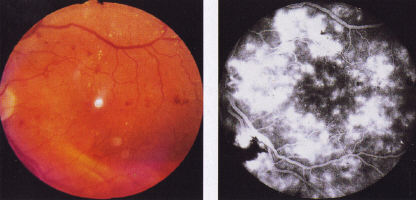
2.4.1.3 Ischaemia maculopathy
Ischaemia maculopathy is almost a diagnosis of exclusion, based on
unexplained visual loss in the presence of a relatively normal looking
macula. Blot haemorrhages in the paramacular region may be indicative
of ischaemic maculopathy. There may be associated haemorrhages and
exudates elsewhere. The exact extent of the ischaemia can only be seen
on fluorescein angiography (Fig 21a, b). It is therefore important that
patients with maculopathy in which ischaemia is suspected are investigated
with this technique. There does not appear to be a direct correlation
between visual acuity Dan the degree of ischaemia.
.
 |
2.4.1.4 Mixed maculopathy
Many cases do not fit exactly into the groups described above. Frequently,
there is combined pathology particularly of diffuse oedema and ischaemia.
Nevertheless, classifying the maculopathy according to its predominant features
is useful form a therapeutic prognostic point of view.